Chemistry
07.05.2022 18:38
241
243
5
Solved by an expert
What is the limiting reagent when 1.5 moles of nitrogen react with 6 moles of hydrogen?
What is the limiting reagent when 1.5 moles of nitrogen react with 6 moles of hydrogen? N2(g)+3H2(g)—> 2NH3(g)
Show Answers
TrueKing184
4,5(38 marks)
Explanation
Given
N2(g)+3H2(g)—> 2NH3(g)
Number of moles of nitrogen = 1.5 moles
Number of moles of hydrogen = 6 moles
Required: Limiting reagent
Solution
For Nitrogen:

For Hydrogen:
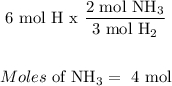
The limiting reagent is Nitrogen, because less moles of NH3 will be produced by nitrogen, meaning it all of it gets used up.
Answer
The limiting reagent is Nitrogen.
jurnee77
4,6(40 marks)
Thanks for the easy points
have a good day
Popular Questions about the subject: Chemistry
Standardized NaOH solutions are typically restandardized if left unused for...
Chemistry
09.06.2022 12:43
If you wanted to travel from Spain to the Caribbean on a sailboat, which winds...
Chemistry
14.09.2021 17:29
The police went on a wild chase to catch this man speeding through town in a...
Chemistry
07.06.2020 18:43
Assuming a mixture of equal volumes of o xylene and cyclohexane,which of these...
Chemistry
10.10.2020 20:53
Hydrogen fuel is clean but not cost-effective. true or false question...
Chemistry
10.08.2022 05:54
What is formula for chromium (iii) phosphate trihydrate and cobalt (ii) phosphate...
Chemistry
12.03.2022 12:15
What element is used in making thermometers...
Chemistry
27.06.2020 14:03
Which values describe standard temperature and pressure (stp)? a. 0°c and 101.3...
Chemistry
11.08.2021 18:37
Ahydrogen-like ion is an ion containing only one electron. the energy of the...
Chemistry
22.07.2020 13:02
Megan prepares a pitcher of lemonade by adding a quarter cup of granular sugar...
Chemistry
26.01.2021 00:50
New questions by subject
Read the passage from President George W. Bush s address to a joint session...
History
07.02.2022 03:31
In the lesson, you explored the idea of themes concerning parent-child relationships...
English
01.03.2020 00:58
Antonie poured 5.6 cups of water into her dogs water bowl this morning. If her...
English
06.03.2023 01:46
JOIN MY MEETING ID-9384026088 PASSWORD 1234...
Biology
22.04.2022 02:37
Choose all the examples that can be measured using volume.? the amount of cereal...
Mathematics
29.03.2022 03:57
Does the relation {(5,-2), (5,-8), (4,-6), (7,-1)} represent a function?...
Mathematics
12.10.2020 08:01
A science class was completing an experiment on electrical circuits. Why did...
Chemistry
22.05.2022 14:12
Pls help Find the radius of a circle whose arc length is 55 m and its central...
Mathematics
22.07.2022 05:27
Which statement best describes Saddam Hussein’s regime? He established a Sunni...
History
29.06.2023 01:06
Zack is creating a four-digit password for his new electronic device. Each digit...
Mathematics
17.07.2022 00:20
Find out answers to questions
from an AI-bot
from an AI-bot
Get full access
Answers






