Why H +ve acts as lewus acid? Why NH3 acts as bronsted-lowery base? Why BF3 acts
Explanation:
Unlike the traditional definition of defining acid as any substance that can react with a base to form salt and water, there are two other definition of acid.
These are lewis acid and bronsted lowry acid.
Lewis acid is any substance that can accept a pair of non bonding electrons. This is why H+ is able to as a lewis acid.
Bronsted lowry definition on the other hand is as follows; A brosted lowry acid is any species that can donate a proton to another molecule. A bronsted lowry base is any specie that can accept a proton from another molecule. NH3 is able to accept a proton, hence it is termed as a bronsted lowry base.
BF3 act as a lewis acid because it can accept accept a pair of non bonding electrons. Generally; an atom, ion, or molecule with an incomplete octet of electrons can act as an Lewis acid.
Sound waves travel faster in a low-density gas
Explanation:
First of all, let's remind that sound waves are pressure waves: they consist of oscillations of the particles in a medium, which oscillate back and forth along the direction of motion of the wave (longitudinal wave).
The speed of sound in an ideal gas is given by the equation
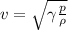
where
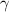 is the adiabatic index of the gas
is the adiabatic index of the gas
p is the gas pressure
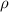 is the gas density
is the gas density
From the equation, we see that the speed of sound is inversely proportional to the square root of the density: therefore, the lower the density, the faster the sound waves.
So, sound waves will travel faster in a low-density gas.
Popular Questions about the subject: Chemistry
New questions by subject
from an AI-bot






