Chemistry
17.08.2021 12:31
258
275
9
Solved by an expert
The question is in the photo (Sorry for being lazy lol )
The question is in the photo (Sorry for being lazy lol )
Show Answers
pinkpearl20
4,9(74 marks)
• When Sodium Sulfate is mixed with Barium chloride, the following reaction occurs :
BaCl2 (aq) + Na2SO4 (aq) > BaSO4 ( s) + 2 NaCl( aq)• a precipitate is the solid that forms, the white solide will be BaSO4
,• So option 4 is correct. BaSo4.
viktoria1198zz
5,0(30 marks)
The mass of NaCl : 50.5 g
Further explanationThe mole is the number of particles(molecules, atoms, ions) contained in a substance
1 mol = 6.02.10²³ particles
Can be formulated
N=n x No
N = number of particles
n = mol
No = Avogadro's = 6.02.10²³
There are 5.20 x 10²³ units of NaCl, so mol NaCl :
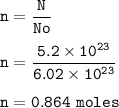
Mass of NaCl(MW=71 g/mol) :
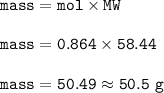
Popular Questions about the subject: Chemistry
Which of the following is NOT an example of a heterogenous solution?...
Chemistry
03.06.2021 15:21
if K & Br were to form a chemical bond, which element would...
Chemistry
21.01.2023 19:04
DOES ANYONE KNOW? I think I know them but I want to be sure...
Chemistry
05.08.2021 03:29
1. Mg(s) + 2HCl(aq) --- MgCl2(aq) + H2(g) a. How many grams of...
Chemistry
23.07.2022 07:43
Why bromine is a ring deactivator as it is ortho-Para directing...
Chemistry
15.09.2021 07:56
calculate the volume of carbon(iv)oxide released when 30dm^3 of...
Chemistry
22.01.2022 11:28
Are plants organisms? Can they invade? Do they compete? What would...
Chemistry
26.08.2020 05:06
Find the mass in grams of 6.02 • 10 ^ 23 particles of NH 3...
Chemistry
24.03.2020 06:17
Gaseous methane (CH4) reacts with gaseous oxygen (O2) gas to produce...
Chemistry
13.05.2023 09:24
Silver sulfadiazine burn-treating cream creates a barrier against...
Chemistry
18.08.2020 03:58
New questions by subject
Which conditional statement is true? If it is a table, it has four...
Mathematics
30.07.2020 06:53
Which statement is true about ionic compounds? A) They are made...
Chemistry
23.04.2020 17:07
What is the output for the following code? Enter the result in...
Computers and Technology
25.04.2020 14:30
HELPP Solve the equation 15+25=4+6x...
Mathematics
12.02.2020 00:24
How did the function move from the f(x)=|x| to f(x)=|x+2|...
Mathematics
01.05.2022 12:25
A 45 gram sample of a substance that’s used to sterilize surgical...
Mathematics
01.03.2023 07:34
I need help plz help me 50 points i will give brainliest if you...
Mathematics
27.08.2022 20:23
Find the slope and y-intercept of the line shown below. A 10 Q...
Mathematics
14.03.2023 18:56
There are at least 15 more football players than baseball players...
Mathematics
15.03.2022 01:58
Some scientists believe Earth s orbit around the sun has changed...
Biology
24.06.2021 22:25
Find out answers to questions
from an AI-bot
from an AI-bot
Get full access
Answers






