¿QUE DATO DE LA CONFIGURACIÓN ELECTRÓNICA TE PERMITE UBICAR LOS ELEMENTOS QUÍMICOS
The question is: WHAT DATA OF THE ELECTRONIC CONFIGURATION ALLOWS YOU TO LOCATE THE CHEMICAL ELEMENTS IN THE PERIODIC TABLE?
Explanation:
Elements which have same number of valence electrons tend to show similar chemical properties due to which they are placed in the same group.
Hence, electronic configuration of an element tells us the number of valence electrons present in an element. As a result, it becomes easy to locate the element in its respective group.
a)0.4222 moles of the compound are present.
b)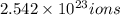 of iron(III)
of iron(III)
c)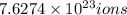 of bromide.
of bromide.
Explanation:
Moles = 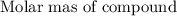
a)Moles of iron (III) bromide =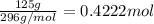
0.4222 moles of the compound are present.
Molecules of of iron (III) bromide in 0.4222 moles
1 mole = 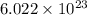 molecules
molecules
In , 0.4222 moles:
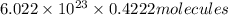
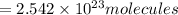 of iron (III) bromide
of iron (III) bromide
b)1 molecule of iron (III) bromide contain 1 iron(III) ion.Then 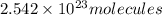 will contain:
will contain:
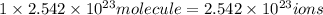 of iron(III)
of iron(III)
c) 1 molecule of iron (III) bromide contain 3 bromide ion.Then 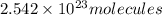 will contain:
will contain:
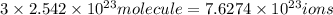 of bromide.
of bromide.
Popular Questions about the subject: Chemistry
New questions by subject
from an AI-bot






